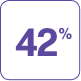Managing hypoparathyroidism
NATPARA works by treating low blood calcium in hypoparathyroidism
NATPARA is the first and only FDA-approved prescription parathyroid hormone that is taken along with calcium and vitamin D to control hypocalcemia in adults with hypoparathyroidism.
PTH balances the level of calcium and vitamin D in the body in 3 major organs by:
Activating vitamin D to help the body get calcium from food
Increasing the amount of calcium the kidneys reabsorb

Releasing calcium from the bones into the bloodstream
NATPARA helps to control your blood calcium level
NATPARA helps raise low blood calcium levels caused by hypoparathyroidism. It works by:
- Helping your intestines absorb calcium by activating vitamin D
- Increasing the amount of calcium your kidneys reabsorb
- Releasing calcium from your bones
NATPARA may cause serious side effects, including:
High blood calcium (hypercalcemia)
-
NATPARA can cause some people to have a higher blood calcium level than normal.
- Your doctor should check your blood calcium before you start and during your treatment with NATPARA.
- Tell your doctor if you have nausea, vomiting, constipation, low energy, or muscle weakness. These may be signs that you have too much calcium in your blood.
NATPARA helped reduce the need for oral calcium supplements and reduced or removed the need for active vitamin D
In a 24-week clinical study in 124 adults with hypoparathyroidism:
-
To prove the efficacy of NATPARA, by the end of the study patients had to have their:
- Dose of active vitamin D reduced by at least 50%
- Dose of calcium reduced by at least 50%
- Blood calcium level in a certain target range (between 7.5 mg/dL and 10.6 mg/dL)
At the end of the clinical study, there were no differences in the percentage of patients with serum calcium levels between 7.5 mg/dL and 10.6 mg/dL in the NATPARA (86.9%) and placebo (87.5%) groups.
Of the 124 adult patients, 84 were taking NATPARA and 40 were taking placebo.
NATPARA may cause serious side effects, including:
Low blood calcium (hypocalcemia)
- People who stop using or miss a dose of NATPARA may have an increased risk of severe low blood calcium levels
- Tell your doctor if you have tingling of your lips, tongue, fingers and feet, twitching of face muscles, cramping of feet and hands, seizures, depression, or have problems thinking or remembering
Tell your doctor right away if you have any of these signs and symptoms of high or low blood calcium levels.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.